July 24, 2013
Asahi Kasei Corp.
Asahi Kasei ZOLL Medical Corp.
Asahi Kasei ZOLL Medical, the Asahi Kasei Group company for critical care devices in Japan, received approval on July 23, 2013, from Japan’s Ministry of Health, Labor and Welfare for the manufacturing and marketing of the LifeVest® Wearable Cardioverter Defibrillator as Designated Marketing Authorization Holder.
Sudden cardiac death (SCD) is the cause of death for approximately 60,000 people per year in Japan. In some cases, it is possible to identify a risk of SCD before it occurs. Being worn continuously (except during bathing), the LifeVest® Wearable Cardioverter Defibrillator provides protection for patients at risk of SCD by constantly monitoring the patient’s heart and, if a life-threatening heart rhythm (ventricular fibrillation or ventricular tachycardia) is detected, delivering a treatment shock to restore normal heart rhythm. The LifeVest® is used to protect a wide range of patients, including patients with newly diagnosed heart failure, following recent myocardial infarction, or following coronary revascularization.
The LifeVest® has one set of electrodes to measure electrocardiogram (ECG) data, and another set of electrodes to deliver the treatment shock. As the detection of life-threatening heart rhythm and delivery of the defibrillation shock are performed automatically, a patient’s life can be saved without assistance of any other person. Furthermore, as the LifeVest® is wearable, its use can be easily initiated or discontinued in accordance with changes in the patient’s condition, enabling use to be limited to the period when a patient is at high risk of SCD.
Outline of the approval
| Approval number: | 22500BZI00017000 | |
|---|---|---|
| Foreign Manufactured Medical Device’s Manufacturing and Marketing Approval Holder: | ZOLL Lifecor Corporation (US) | |
| Designated Marketing Authorization Holder: | Asahi Kasei ZOLL Medical Corp. |
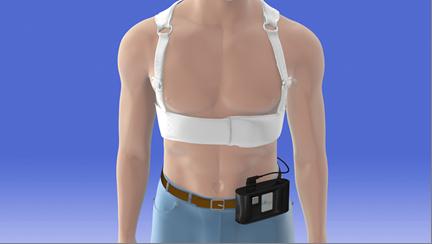
Illustration of LifeVest® being worn
About Asahi Kasei ZOLL Medical
Asahi Kasei ZOLL Medical (AZM), a subsidiary of Asahi Kasei Group company ZOLL Medical Corporation, was established in August 2012 to sell ZOLL critical care products in Japan. AZM will continue to seek regulatory approval for other ZOLL products for their sale in Japan, while working to achieve synergy with the other health care operations of the Asahi Kasei Group.
Adobe Readeris required to view these PDF files.