July 30, 2014
Asahi Kasei Corp.
Asahi Kasei ZOLL Medical Corp.
Asahi Kasei ZOLL Medical (AZM), the Asahi Kasei Group company for acute critical care devices in Japan, received approval on June 30, 2014, from Japan’s Ministry of Health, Labor and Welfare for the manufacturing and marketing of the R Series® defibrillator as Designated Marketing Authorization Holder.
Manufactured by AZM’s parent company ZOLL Medical Corporation (ZOLL), the R Series® is a defibrillator that includes monitoring functions for use in hospital operating rooms, intensive care units, etc. It is the leading defibrillator in the US hospital market. In addition to defibrillation, it provides external pacing as well as monitoring of electrocardiograph (ECG), non-invasive blood pressure, pulse oximetry (SpO2), and end-tidal carbon dioxide concentration (EtCO2).
The R Series® also incorporates the Real CPR Help® function, a proprietary technology of ZOLL. This provides real-time feedback on rate and depth of compressions, helping rescuers perform higher-quality cardiopulmonary resuscitation (CPR). Its See-Thru CPR® function furthermore filters out compression artifact on the ECG monitor so that rescuers can see the underlying heart rhythm during CPR, thereby reducing the duration of pauses in compressions.
AZM is now advancing preparations for sale of the R Series® in Japan, with launch planned for spring 2015.
Outline of the approval
| Approval number: | 22600BZI00005000 | |
|---|---|---|
| Foreign Manufactured Medical Device’s Manufacturing and Marketing Approval Holder: | ZOLL Medical Corporation (US) | |
| Designated Marketing Authorization Holder: | Asahi Kasei ZOLL Medical Corp. |
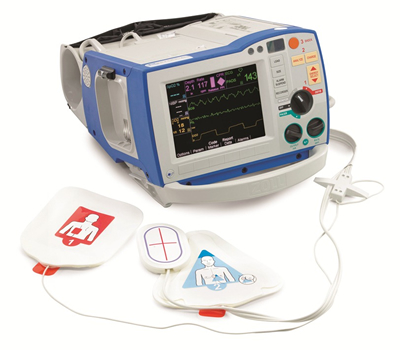
R Series® defibrillator
About AZM
AZM was established in August 2012 to sell ZOLL acute critical care products in Japan. AZM is working to expand its contribution to the field of acute critical care in Japan, while achieving synergy with the other health care operations of the Asahi Kasei Group.
Adobe Readeris required to view these PDF files.