- Food
- Separation technology
Contributing to increasing productivity in the separation and filtration processes of the food industry from both the software and hardware sides
contents list
Microza responds to various challenges in the food application
Automate the filtration process to reduce job labor and save manpower
Lessen the physical load on microorganisms during the purification process
Improve the separation precision to meet the required quality standards
Cut costs and energy consumption the separation process
Decrease the amount of wastes from the filtration process
Why Microza?

Extensive product lineup
Microza products are available in a variety of membrane materials, separation precisions, and hollow fiber inner diameters. For food applications, there are over 20 types in the lineup , including modules for small-scale testing and commercial-scale modules.
Low leakage risk of the target substance
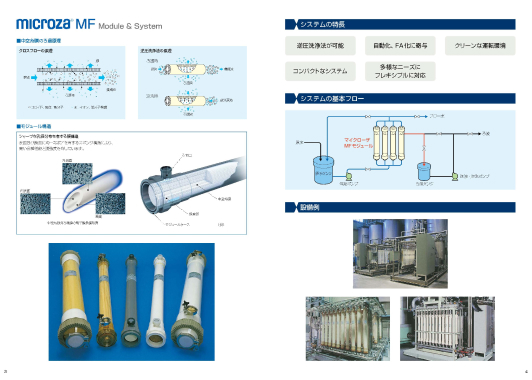
Separation layers are formed on both the inner and outer sides of the Microza hollow fiber (UF) membrane. The original double-skin structure can reduce the risk of leakage of the target substance. “Pressurized backwashing” is also possible where the pressure on the permeate is instantaneously reversed from the permeate side (secondary side) to the feed side (primary side), causing the permeate to flow backward and preventing clogging.

Extensive track record
Microza products cover a wide range of applications, such as carbohydrates, alternative proteins, honey, amino acids, sake, health food raw materials, enzymes, soy sauce, lactic acid bacteria, algae, cultured meat, and extracts. We propose the Microza product optimum for your production process.

High durability and chemical resistance
Microfiltration (MF) membranes that are made of polyvinylidene difluoride (PVDF), which is fluorine-based resin, have strong mechanical properties and excellent durability and chemical resistance. Like ultrafiltration (UF) membranes, they can also be back washed.

No need to purchase filtration aid
Microza can reduce the costs of the separation process because it will be unnecessary to purchase filter aid or dispose of them.

Enabling automated and manpower-saving operation
Filtration completes just by turning on the switch, and thus the automated and manpower-saving separation process improves work efficiency. The modules need not be disassembled or washed after use, and washing completes by circulating chemicals within the device. It is also feasible for nighttime operations.

High filtration stability even at a low linear velocity
Selection of optimum filtration conditions enables separation and concentration to be performed by imposing fewer loads on microorganisms.
Our Products
Pencil-scale and laboratory-scale testing modules
Small-scale hollow fiber membrane modules for testing samples of small volumes

Ultrafiltration membrane: AP and SP series
AP and SP series are ultrafiltration (UF) membrane modules of high sanitation performance and are widely used in food, pharmaceutical, and bioprocess applications. Both circulation and filtrate-side nozzles are connected to the pipes of the filtration systems with ferrules.

Microfiltration membrane
Microfiltration (MF) membranes of highly durable polyvinylidene difluoride (PVDF) and polyethylene (PE) that apply to a wide range of pH are available.
Case studies
- Health food company A
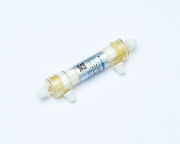
Improving the productivity of concntration and purification process of lactic acid bacteria
Cultured lactic acid bacteria are collected, and the low-molecular-weight fraction originating from the culture medium is removed by filtering while adding water (washing of the cell bodies).
The manufacturer wanted to increase the productivity in bacteria collection to respond to increases in demand for the product but faced a problem with the existing membrane facility requiring too much treatment time.To solve the problem, Microza MF membrane, which manifests a high recovery rate of lactic acid bacteria and has a large membrane area, was introduced. It resulted in an increased capacity of bacteria recovery rate even in a limited installation space.Membrane filtration is easy to scale up, and thus MF membranes have been widely selected for scaling up from a small centrifuge.
- Starch sugar company A
Cost and waste reductions in the purification process of starch sugar

Removal of raw material-derived solids and aggregated proteins after the saccharification process
In the production of sugar from starch, product purity after the saccharification process is increased by removing suspended matter and coloring components. The manufacturer realized that the conventional diatomite filtration required huge costs for filter aid and is not environment-friendly as a huge amount of industrial waste is produced. Implementation of Microza MF membrane into the clarification process not only improves the economy by reducing the costs of filter aid but also reduces industrial waste. It is very burdensome to change the production process which has a long history, but Asahi Kasei accompanies and supports customers in investigating changes.
- Health food company B
Productivity improvement of enzyme production

1) MF membranes to collect and purify microorganisms after culture/fermentation
2) UF membranes to concentrate and recover enzyme after MF
There are several ways to purify enzymes produced by microorganisms. Out of them, microorganism collection and clarification using Microza MF membrane are highly work efficient and achieve economic merits. MF membrane having a large hollow fiber inner diameter is also feasible for removing solid culture media components. For downstream enzyme concentration, a highly hydrophilic polyacrylonitrile UF membrane is proposed to provide a wide range of support for the purification and concentration processes. MF and UF membranes have been widely implemented in the production of enzymes not only for sales but also for in-house use.