- Features
- Performances
- Specifications
- References
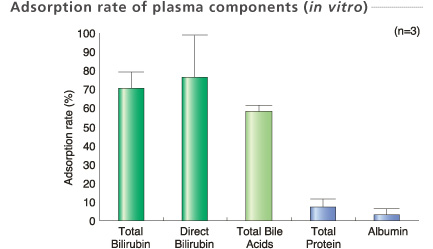
Plasma sample were collected at the column outlet point when 2.4 L (equivalent volume) plasma was treated.
Experimental conditions
Perfusion method: One-pass operation with small scale column (1/350)
Sample : Human plasma 2.4 L (Equivalent volume)
Flow rate : 20 mL/min (Equivalent rate)
In-house data
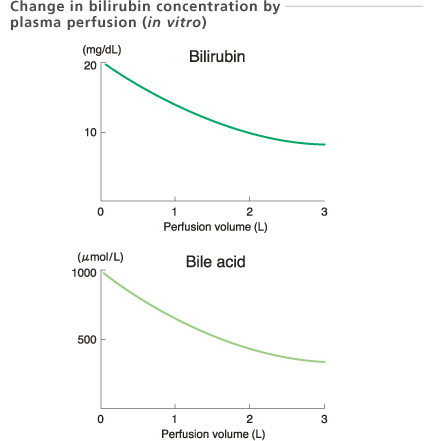
Experimental conditions
Sample : Bovine plasma 2 L (5 units/mL heparin, 20 mg/dL bilirubin and 1000 μ mol/L bile acid added bovine plasma)
Temperature : 37 ℃
Perfusion rate: 20 mL/min
In-house data
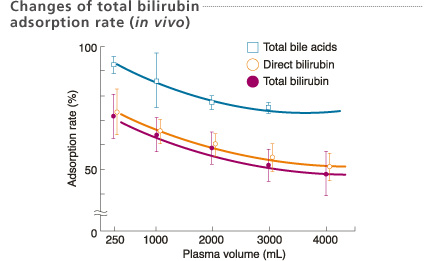
Patients : n=6
Total treatments : 29 times
First Department of Surgery, Okayama University, Japan
Trademark
PLASORBA is a trademark of Asahi Kasei Medical Co., Ltd.

PLASORBA BR
Please feel free to send us any questions you may have about our products and support.
Share your feelings and experiences when using our products.