- Features
- Performances
- Specifications
- References
- Hirata et al.
- IMMUSORBA TR and PH.
- Ther Apher Dial. 7:85-90, 2003.
- Yoshida et al.
- IMMUSORBA TR and IMMUSORBA PH: Basics of design and features of functions.
- Ther Apher. 4:127-34, 2000.
- Yoshida et al.
- IMMUSORBA TR and IMMUSORBA PH: Basics of design and features of functions.
- Ther Apher. 4:127-34, 2000.
- Haupt et al.
- Apheresis and selective adsorption plus immunoglobulin treatment in Guillain-Barré syndrome.
- Ther Apher. 4:198-200, 2000.
- Ota et al.
- Neutrophil activation in immunoadsorption.
- Ther Apher. 4:229-34, 2000.
- Yang et al.
- Plasma adsorption in critical care.
- Ther Apher. 6:184-8, 2002.
- Hirata et al.
- IMMUSORBA TR and PH.
- Ther Apher Dial. 7:85-90, 2003.
- Beige et al.
- Immunoadsorption with tryptophan adsorbers for successful treatment of late steroid-refractory recurrent focal glomerulosclerosis.
- Am J Transplant. 3:1459, 2003.
- Splendiani et al.
- Plasmaperfusion on triptophan columns can improve the clinical outcome of patients affected with myasthenia gravis.
- Artif Cells Blood Substit Immobil Biotechnol. 31:69-79, 2003.
- Luftl et al.
- Successful removal of pathogenic autoantibodies in pemphigus by immunoadsorption with a tryptophan-linked polyvinylalcohol adsorber.
- Br J Dermatol. 149:598-605, 2003.
- Okamiya et al.
- Tryptophan-immobilized column-based immunoadsorption as the choice method for plasmapheresis in Guillain-Barre syndrome.
- Ther Apher Dial. 8:248-53, 2004.
- Seta et al.
- Factors influencing outcome in Guillain-Barré Syndrome: comparison of plasma adsorption against other treatments.
- Clin Neurol Neurosurg. 107:491-6, 2005.
- Arakawa et al.
- Immunoadsorption therapy for a child with Guillain-Barré syndrome subsequent to Mycoplasma infection: a case study.
- Brain Dev. 27:431-3, 2005.
- Wagner et al.
- [Long-term treatment of refractory myasthenia gravis with immunoadsorption].
- Dtsch Med Wochenschr. 133:2377-82, 2008. German
- Wenning et al.
- Treatment of progressive multifocal leukoencephalopathy associated with natalizumab.
- N Engl J Med. 361:1075-80, 2009.
- Galldiks et al.
- Immunoadsorption in patients with chronic inflammatory demyelinating polyradiculoneuropathy with unsatisfactory response to first-line treatment.
- Eur Neurol. 66:183-9, 2011.
- Nagatomo et al.
- Specific immunoadsorption therapy using a tryptophan column in patients with refractory heart failure due to dilated cardiomyopathy.
- J Clin Apher. 26:1-8, 2011.
- Nagane et al.
- Early aggressive treatment strategy against myasthenia gravis.
- Eur Neurol. 65:16-22, 2011.
- Köhler et al.
- A randomized and controlled study comparing immunoadsorption and plasma exchange in myasthenic crisis.
- J Clin Apher. 26:347-55, 2011.
- Trebst et al.
- Immunoadsorption therapy for steroid-unresponsive relapses in patients with multiple sclerosis.
- Blood Purif. 33:1-6, 2012.
- Koziolek et al.
- Immunoadsorption therapy in patients with multiple sclerosis with steroid-refractory optical neuritis.
- J Neuroinflammation. 26;9:80, 2012.
- Koziolek et al.
- Immunoadsorption in steroid-refractory multiple sclerosis.
- Atheroscler Suppl. 14:175-8, 2013.
- Klingel et al.
- Neurologic diseases of the central nervous system with pathophysiologically relevant autoantibodies--perspectives for immunoadsorption.
- Atheroscler Suppl. 14:161-5, 2013.
- Heigl et al.
- Immunoadsorption in steroid-refractory multiple sclerosis: clinical experience in 60 patients.
- Atheroscler Suppl. 14:167-73, 2013.
- Yonee et al.
- Association of acute cerebellar ataxia and human papilloma virus vaccination: a case report.
- Neuropediatrics. 44:265-7, 2013.
Trademark
IMMUSORBA is a trademark of Asahi Kasei Medical Co., Ltd.

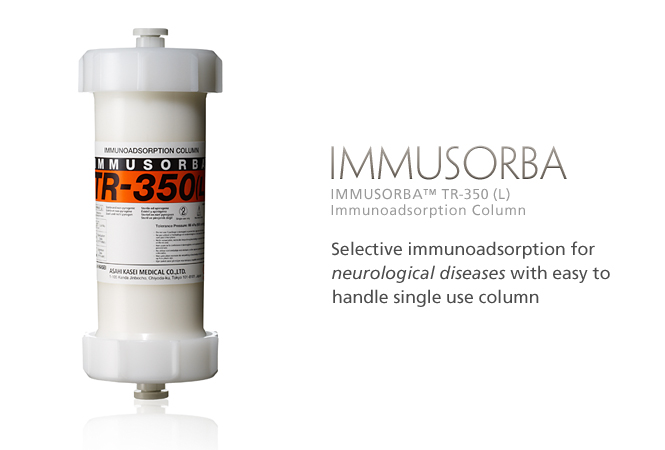
IMMUSORBA TR
Please feel free to send us any questions you may have about our products and support.
Share your feelings and experiences when using our products.